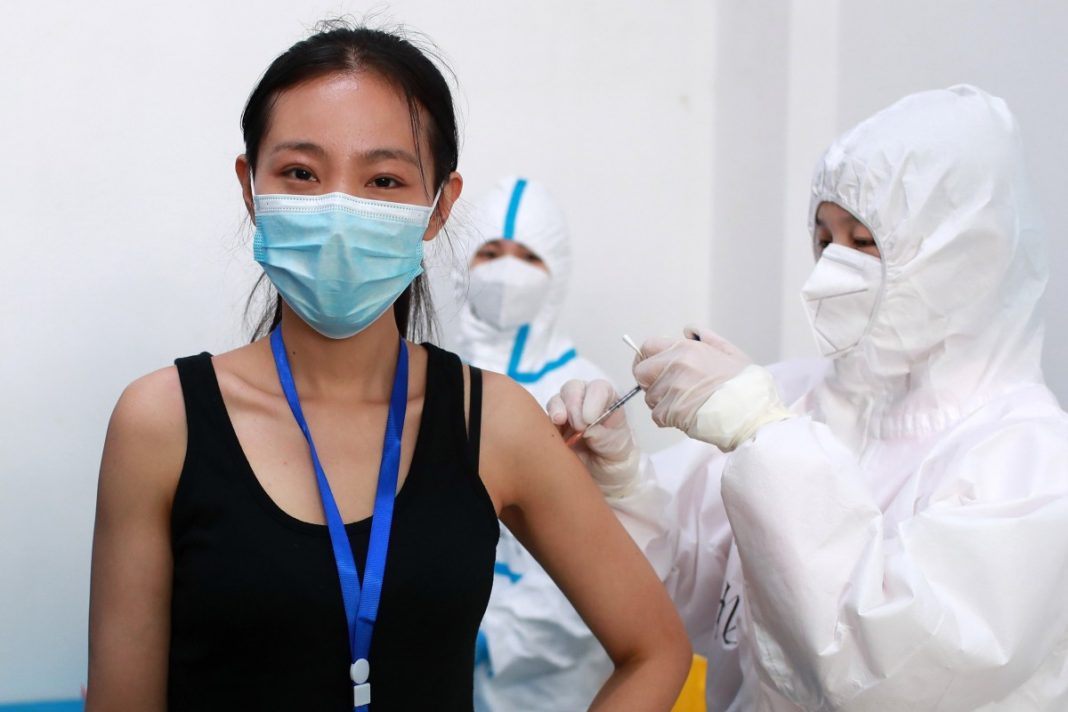
Since the Coronavirus Pandemic hit the world in 2020 we’ve seen exciting developments unfolded by many countries regarding Covid-19 drugs.
Many governments working on the situation given us hope with the success news of vaccine trials and drug improvements. Unfortunately, we could rely on very few of them.
Why the question for reliability came forward in the time when humanity needs a cure for the pandemic?
Experts say that most of the drugs and vaccines approved by different governments had little or no scientific backup. For instance, the U.S government approved a malaria drug to treat Covid-19 patients. But is it dependable to cure millions of infected patients?
Before the questions can be answered properly Russia launched its own version of vaccines. Again it was not backed up by scientific studies.
We know the importance of time for developing a possible cure for a pandemic. But it doesn’t mean providing quicker but not tested drugs instead of the tested ones.
So are we approving the Covid-19 drugs and vaccines very easily? What will be the consequences of developing early solutions?
- More Stories: Australian Reporters Removed from China due to Diplomatic Standoff
- More Stories: Bill Gates Sees the Future in a Korean Vaccine
Hydroxychloroquine Used as Covid-19 Drugs
America’s Food and Drug Administration (FDA) issued a malaria drug to cure Covid-19 patients in March 2020. The drug is called Hydroxychloroquine trusted by many during the time.
Unfortunately, the drug is not risk-free. There is no evidence that it can cure the virus inside our bodies. Still many people including President Donald Trump recommended the drug to the world.
The drug was authorized based on a “limited in vitro and anecdotal clinical data.”
The move was fast but not safe to be the possible solution for a quicker recovery. And finally, the drug was canceled later since it had little effect on the Covid-19 patients.
Rather than being a possible solution, Hydroxychloroquine actually killed the time for us in the search for Covid eradication.
Russian Vaccine and Trump’s Response with Plasma Therapy
Russian President Vladimir Putin announced on August 11th that his government approved the first Covid-19 vaccine in the world.
But the lack of proper scientific research again created doubt in our minds. Experts who have been watching the failure of the Hydroxychloroquine trial and failure as a drug now think Sputnik V will have the same fate.
Once again the U.S authorities approved another non-scientific approach by approving plasma therapy. The announcement came on August 23rd and President Trump said the therapy as a “very historic breakthrough.”
However, concerns over the proper testing procedures still exist for Plasma Therapy.
In concluding remarks, we can say that a quick but tested approach should be taken to develop Covid drugs and vaccines. If not done properly we will make a fundamental mistake that can’t be overturned.
Read More: Severe Remittance Loss Due to Covid-19 Might distress the Asia Pacific Economy